In addition to cutting cost and saving time, there are a plethora of benefits in implementing ISO 13485 2016 QMS standard: Focus on product cleanliness requirementsīenefits associated with ISO 13485 include:.Increased focus on the control of suppliers.Development & improvement on many regulations.Clarification on training responsibilities.Clarification on management responsibilities.In March 2016, the ISO 13485 standard was updated. The other prominent updates include the changes in regards to the increased regulatory requirements for the supply chain industries.
#RISK MANAGEMENT ISO 13485 UPDATE#
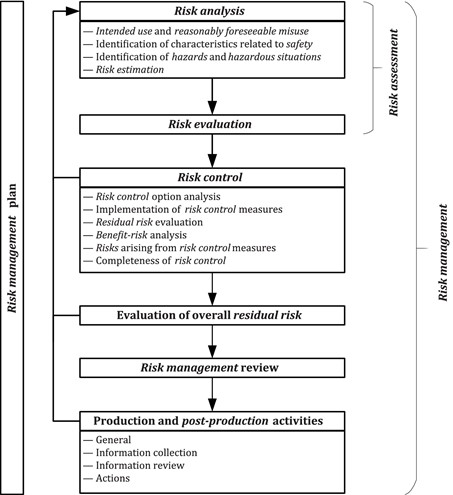
The primary update is the increased focus on risk management and risk-based decision-making. The new standard is compatible with all technological advancements along with the regulatory requirements and also meets other expectations. The standard has been updated to reflect the most up-to-date quality management system techniques.
#RISK MANAGEMENT ISO 13485 FULL#
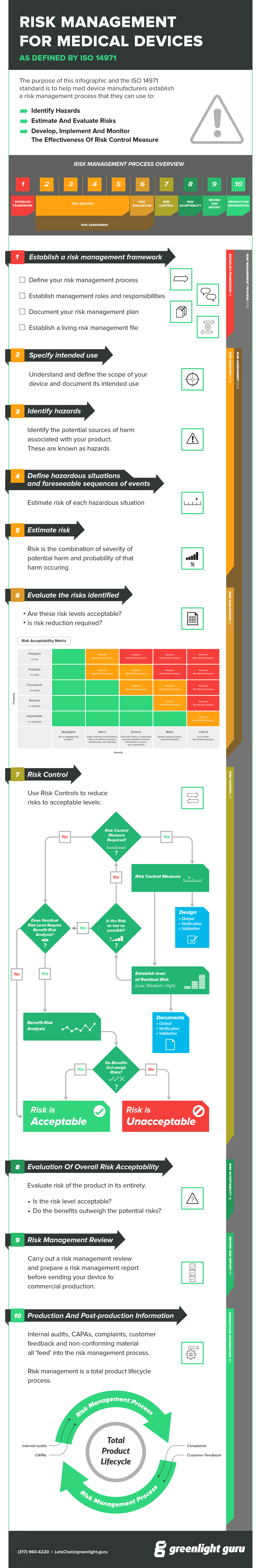
Section 5–Management Responsibility –Implementing and maintaining and planning QMS, review and ensure customer satisfaction.Section 4 – Quality Management System – General QMS, documentation, and quality control requirements.The ISO 13485 standard is divided into 8 sections the first 3 are introductory and the last 5 are the necessary and important requirements of the QMS. Having a certificate of ISO 13485 will add value to the Organisation and assure trust for customers. Key requirements of ISO 13485?Ĭertification to the ISO 13485:2016 QMS standard is not necessarily a requirement of the ISO 13485 however, companies can benefit from the standard without actual certifying to the ISO 13485 standard. A summary of the changes incorporated into this edition compared with the previous edition is given in Annex A. Technical Corrigendum ISO 13485:2003/Cor.1:2009 is also incorporated in it. The second edition (ISO13485:2003) and ISO/TR 14969:2004 have been technically canceled and got replaced with the third edition (ISO 13485). Along with the medical device industries, the entire medical device supply chain also needs to adhere to the specifications of the ISO 13485 standard. The standard was revised in 2003 wherein all the documents were combined into one that paved way for one main standard that could be universally applied for all medical device manufacturers. ISO 13488 for manufacturers on contract or for the suppliers.Initially, this standard (ISO 13485) was published in two versions: It was first published in the year 1996 by the International Organization for Standardization (ISO). It primarily demonstrates the ability of an organization to provide quality medical devices and to fulfill customer and regulatory requirements. The requirements for quality management systems are specified in ISO 13485:2016. This standard was established to provide medical device companies with requirements to ensure and maintain quality systems.

The International Organization for Standardization (ISO) initially released the ISO 13485 Quality Management System Standard in 1996.
